I’ve been struggling to understand how water vapor affects tire pressure. So I launched an effort to do so. There were a number of questions:
- Does water vapor behave like an Ideal Gas?
- Answer: Yes. Water Vapor, like most gases, behaves like an Ideal Gas at the temperatures and pressures we deal with in tires.
- How do you handle the liquid water going in and out of phase (liquid-to-gas / gas-to-liquid) inside a tire as it heats up and cools down during normal operation?
- See below
- Does the fact that bottled nitrogen normally does not contain water vapor have appreciable effect on tire pressure?
- See below
When is water a gas and when is it a liquid?
Let’s start with the most important bit of information needed: How do we determine when water is a gas vs when it is a liquid?
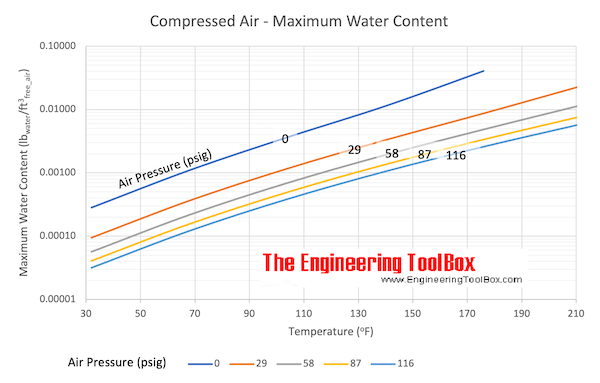
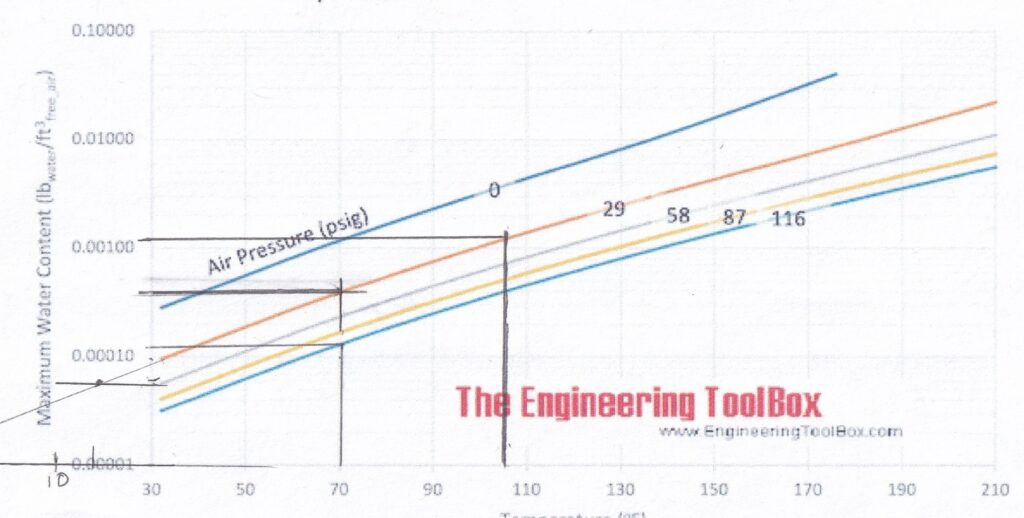
This is a graph of Saturated Air (100% humidity) for various Pressures and Temperatures. Please note that the odd increments for pressure is due to this graph being a translation of one done in metric units.
Zero pressure means ambient pressure = 14.7 psia (101 kPa). All those lines are what you would read on a pressure gauge.
Anything below those lines would cause some of the water to precipitate out (rain!) until the air is holding as much water vapor as it can = 100% relative humidity.
For those who don’t remember, as the temperature increases, air can hold more water as vapor. That means that if the temperature increases and no water is available to evaporate, the relative humidity goes down.
Please note: It is common for technical folks to refer to absolute pressure by adding an “a” to the end of the pressure units = psia, kPa(a), or kPa abs, or even bar(a). By contrast, for gauge pressure, the letter “g” is sometimes added to the end of the units = psig, kPa(g), kPa gauge, bar(g). HOWEVER, it is much more common for gauge pressure not to have any special notation at all! = psi, kPa, bar.
Now onto the analysis:
If we take an air compressor tank at 116 psi (800 kPa), the air in that tank is typically saturated (100% humidity) at 70°F (21°C), the maximum water content is about 0.00013 lb/cubic foot (0.0021 kg/m3). See the black lines below:
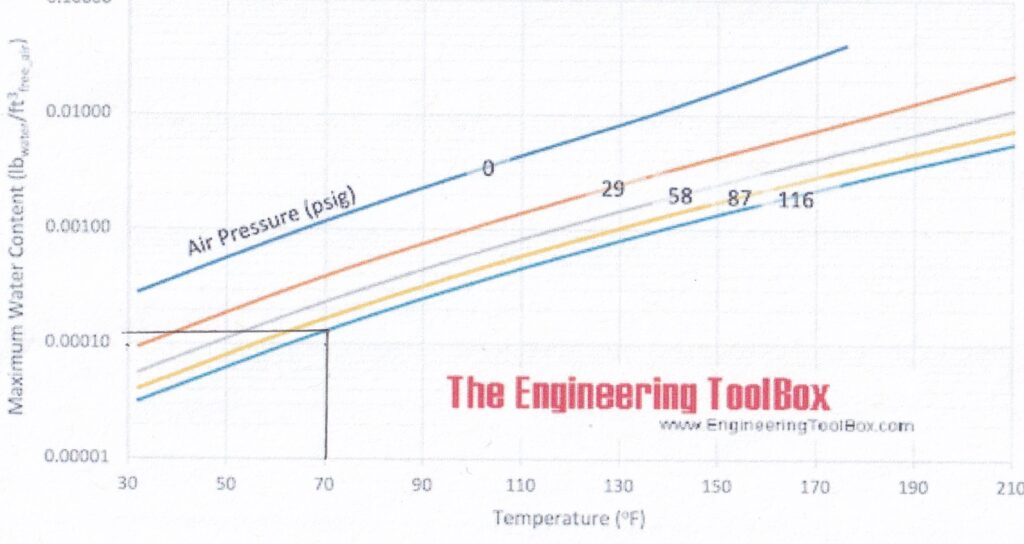
Please note that the scale on Y axis (the left of the graph) is logarithmic = the major lines are a factor of ten (10) more that the next increment down.
Air behaves very much like an Ideal Gas in these conditions. So if I fill a tire to 29 psi (200 kPa), then the amount of water vapor in the tire is changed by the fraction of the absolute pressures:
(116+15) / (29+15) = 131 / 44 ≈ 3
That means the water content is about a third of what it was in the tank – about 0.00004 lb/cubic foot (0.00064kg/m3) – which makes the 100% humidity point (Dew Point) about 18°F (-8°C) Note: I had to extrapolate from that graph. See below.
And that is below the freezing point of water at that pressure, so the water condenses out as frost (snow!)
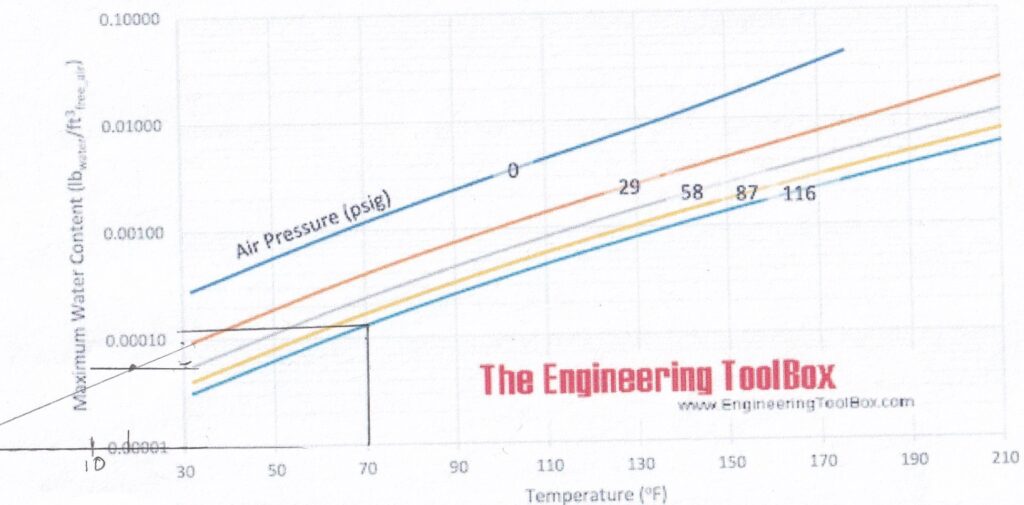
But the important thing is that at 70°F (21°C), the tire, when inflated to 29 psi (200 kPa), is inflated with air that is less than 100% relative humidity. It is not adding liquid water to the tire, nor is water condensing out during the inflation process.
And just an FYI, if I start at 70°F and 29 psi, the normal max temperature/pressure the air chamber a street tire should reach is 106°F / 32 psi. That’s based on a rule of thumb of a 10% pressure increase. Please note, that is the average temperature of the air chamber, not the temperature of the tread surface.
Since 100% of the air mixture in the tire’s air chamber starts out as a gas, it will follow the Ideal Gas Law.
Let’s try it when there is a pool of water inside the tire!
For this case, the air inside the air chamber is saturated with water vapor (100% humidity), but that gaseous mixture behaves more or less like an Ideal Gas. As the tire operates, it generates heat and some of the liquid water will “evaporate” (I’m sure there is a better word for this process and “Boiling” doesn’t seem like the word to use!!) . Here’s how to calculate that:
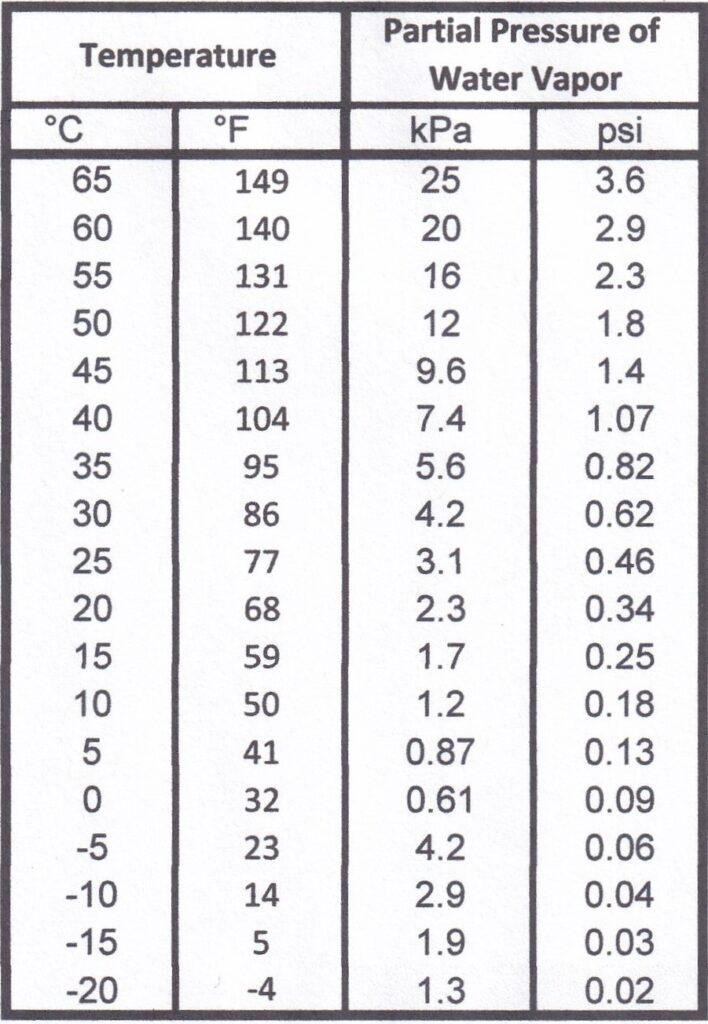
This chart is the partial pressure of water vapor at various temperatures for fully saturated air. The partial pressure of water vapor is very much tied to temperature and not to the pressure of the gaseous mixture.
Note: I’ve only included temperatures we are going to encounter in a tire’s air chamber.
Step 1) Find the starting temperature. Record the partial pressure.
Step 2) Using the Ideal Gas law and the starting pressure and temperature, calculate the operating temperature, and record the corresponding partial pressure.
Step 3) Subtract the 2 values to get the added pressure.
In our Example:
The starting temperature was 68°F (20°C) and 29 psi (200 kPa), and the ending pressure was 32 psi (221 kPa), making our ending temperature 106°F (41°C).
The partial pressure of water vapor at 68°F (20°C) is 0.34 psi (2.3 kPa), and the ending temperature of 106°F is about 1.07 psi (7.4 kPa), making the difference 0.73 psi (5.1 kPa).
For a street tire, that’s OK. For a race tire, that is OK only if there is still water inside at racing temperatures. Please note: A race tire is very likely to run hotter than 106°F (41°C). – remembering we are talking about the air chamber pressure/temperature, not the tread surface!
HOWEVER, the increased pressure is going to cause the tire to operate at a slightly lower temperature (less deflection!). From both the street tire’s and race tire’s point of view, that would be a good thing!
How much water are we talking about?
That is, the amount of starting liquid water inside the tire so that there will always be liquid water inside the tire?
For that we need to know how much more water (in terms of weight/volume, not pressure) and how much volume of saturated air there is.
The amount of water can be obtained from those graphs at the beginning of this article. It’s the difference between 70°F (21°C) and 106°F (41°C) on the 29 psi (200 kPa) line. (Those 2 dark lines!): 0.00110 – 0.00035 = 0.00075 lb/ cubic feet (0.0021 kg/m3)
Now we need the volume of water. To do that we need to consider the tire and wheel as a toroid (doughnut shaped!). The formula for the volume of a toroid is:
V = 2π2R(a*b) – where:
- R = radius of the center of the toroid
- In other words, the rim diameter, plus the section height
- a = width of the toroid
- in other words, the section width
- b = height of the toroid
- In other words, the section height
- Please note: This is a ballpark calculation. I am ignoring that the tire itself has some thickness and it only slightly looks like a toroid – close enough is close enough!!
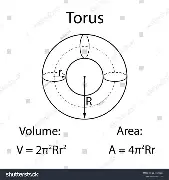
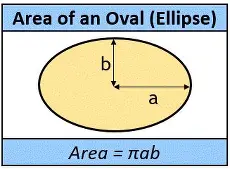
I don’t know why I couldn’t find an image of a toroid with an oval cross section, so I picked 2 images with the formulae – recognizing that a torus has a circular cross section where the area = πr2 – and combined them in the formula!!
ah ……. Mmmm …… So let’s pick a tire size …… ah …… how ’bout 225/45R17 ?
Doing the math: Volume = 8.47 cubic feet (0.23 m3)
To get weight of liquid water multiply by the amount of water in lb/cubic feet = 0.00075 * 8.47 = 0.0063#. (0.0029 kg)
A gallon of water weighs 8.33# (3.78 kg), so that means about a half a teaspoon (2 1/2 mL) of liquid water in the tire is required to ensure we always have liquid water inside the tire!!
What about when the liquid water completely “evaporates” before the tire reaches its operating temperature/pressure?
Now let’s cover the situation between the 2 situations above – the liquid water inside the air chamber at the start is completely “evaporated” as the tire reaches its operating temperature/pressure.
In a street tire, this situation doesn’t have much effect on how the tire behaves. A fraction of a psi will not have a large effect on treadwear, handling, ride, etc.
This is not the case for a race tire, as the race tire’s spring rate is a function of the inflation pressure – and spring rates affect the car’s handling. I’ve heard that NASCAR team use increments of a quarter (1/4) a psi!! (1.7 kPa)
One last item:
Rubber is somewhat porous – that is, it is not completely air tight. The rate of leakage is pretty low, but even water vapor will leak out – at a rate proportional to the partial pressure. That means that eventually the inside of the tire becomes completely dry!!
Summary:
- Filling a tire from a tank results in the tire’s air chamber having REDUCED humidity – that is, not near 100% relative humidity.
- Air – even humid air – behaves like an Ideal Gas.
- That means that if the tire does not have liquid water inside to begin with, the pressure buildup is predicted by the Ideal Gas Law.
- If there is sufficient liquid water at ambient starting temperatures, an inflated street tire will experience about 3/4 of a psi (1.7 kPa) MORE pressure buildup. This is insignificant for a street tire.
- If there is some liquid water – we’re talking less than a half a teaspoon – but not enough to still be liquid water after the warmup phase, the handling of a racecar can be affected and in an unpredictable way. This validates the adage that nitrogen is better than air for race tires.
Final Thoughts:
In this whole article, I’ve referred to a tank type air compressor. But those aren’t the only kind. Here’s one:

This one is for sale at Tire Rack: VIAIR 85P Compressor Kit | Tire Rack
Please note: I am not endorsing this. It’s just one that was easy to find!
There is nowhere for the condensed water to go except through the hose into the tire. For a street tire, that is not a problem – see above. But I want folks to be aware.
